What a Week of Real-World Walking Data Reveals About Parkinson's Disease
Clinical trials in Parkinson's disease have a measurement problem that practitioners feel every day. The MDS-Unified Parkinson's Disease Rating Scale captures how a patient moves during a structured clinic visit. It is carefully standardized, widely understood, and limited to a single 30-minute window. For a disease that produces fluctuating motor states, wearing-off cycles, and variable daily function across thousands of waking hours, a snapshot taken in optimal conditions tells an incomplete story.
The Mobilise-D consortium was established to address this directly. Their recently published validation study brings together a remarkable group: Alison Yarnall and Lynn Rochester at Newcastle, Walter Maetzler, Alice Nieuwboer, Jeffrey Hausdorff, Anat Mirelman, and many others whose work in gait and mobility has shaped the field. It is one of the most careful and detailed real-world mobility characterizations in Parkinson's disease to date. The work reflects years of rigorous methodological development, and the dataset they have assembled will support the field for some time.
The study enrolled 601 individuals with Parkinson's disease and 232 age- and sex-matched healthy controls at sites in Newcastle, Kiel, Erlangen, Leuven, and Tel Aviv. Participants wore a single inertial measurement unit on their lower back continuously for seven days, day and night, capturing movement at 100 Hz. Two validated devices were used across sites (the McRoberts MoveMonitor+ and the Axivity AX6), with no clinically meaningful differences between them in the digital mobility outcomes they produced.
The 24 digital mobility outcomes span four domains: walking activity (how much and in what pattern people walk each day), gait pace (walking speed, stride length), gait rhythm (cadence, stride duration), and bout-to-bout variability. Valid weekly-level data were available for 531 PD participants and 221 controls (88.4% and 96.1% of each cohort respectively). Data loss in PD was largely technical rather than related to poor adherence.
The PD cohort was mild to moderate: mean age 65.7 years, mean disease duration 6.7 years, mean MDS-UPDRS III score 26.3, with 67% at Hoehn and Yahr stage II and 15% at stage III. Eighteen percent were freezers. Over a third reported at least one fall in the preceding year.
Twenty of the 24 digital mobility outcomes showed statistically significant differences between PD participants and controls. The radar plot below, reproduced from the paper, shows the magnitude of those differences across all four domains. The gap between the PD (grey) and control (black) profiles is visible across nearly every spoke.
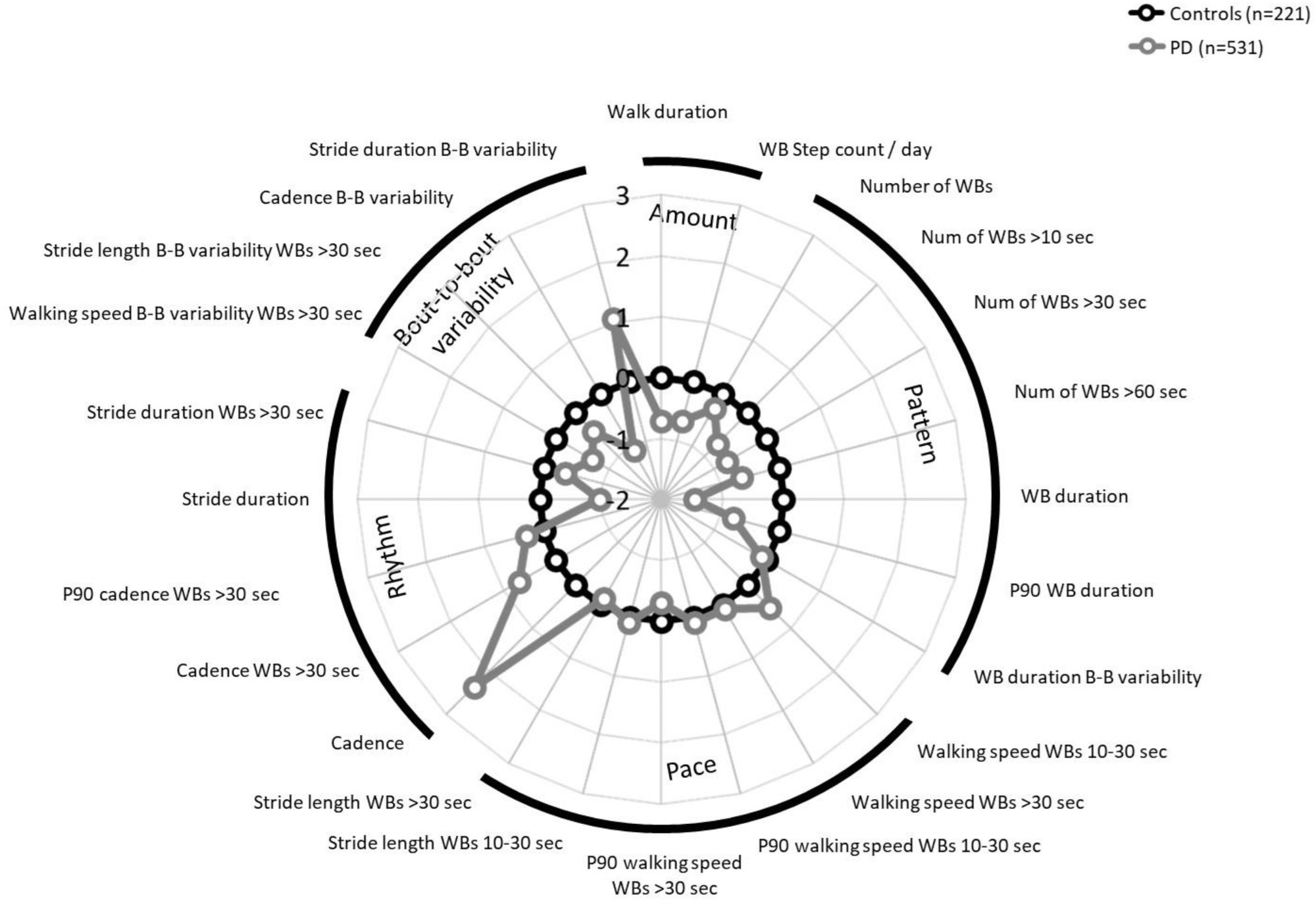
Figure 2 from Yarnall et al. Radar plot of digital mobility outcomes in PD (n=531) vs. controls (n=221). Grey = PD, black = controls. Reproduced under CC BY-NC 4.0.
The walking activity findings were consistent and expected in direction. PD participants walked for a median of 1.35 hours per day, compared to 1.87 hours in controls. They accumulated 7,733 walking bout steps per day versus 10,563 in controls. Their walking bouts were shorter in duration (median 7.80 seconds versus 9.23 seconds) and they had fewer bouts sustained beyond 10, 30, and 60 seconds. The picture is one of fragmented, abbreviated daily mobility.
The gait rhythm findings were also clear. PD participants walked at a higher cadence: 92.1 steps per minute in all walking bouts compared to 84.6 in controls. Stride duration was correspondingly shorter. This is the well-characterized Parkinsonian pattern of rapid, short stepping.
Two findings deserve closer attention because they are less intuitive.
First, in shorter walking bouts (10–30 seconds), PD participants walked faster than controls: 0.74 m/s versus 0.70 m/s. This reversal did not persist in longer bouts, where the groups were comparable. The authors attribute this to compensatory power bursts that PD patients generate to overcome bradykinesia and motor initiation difficulties at the start of walking, an effect that diminishes as bouts lengthen. Walking speed in shorter bouts may therefore reflect motor compensation more than motor impairment, and interpreting it without that context would be misleading.
Second, bout-to-bout variability was lower in PD than in controls across walking speed, stride length, and cadence measures. Healthy walkers naturally adapt their gait to terrain, attention, and context, producing regular variation between bouts. PD patients exhibited more rigid, stereotyped gait patterns between bouts. Lower variability here is a sign of reduced adaptability, not greater precision.
The disease severity analysis is where the clinical trial relevance becomes direct.
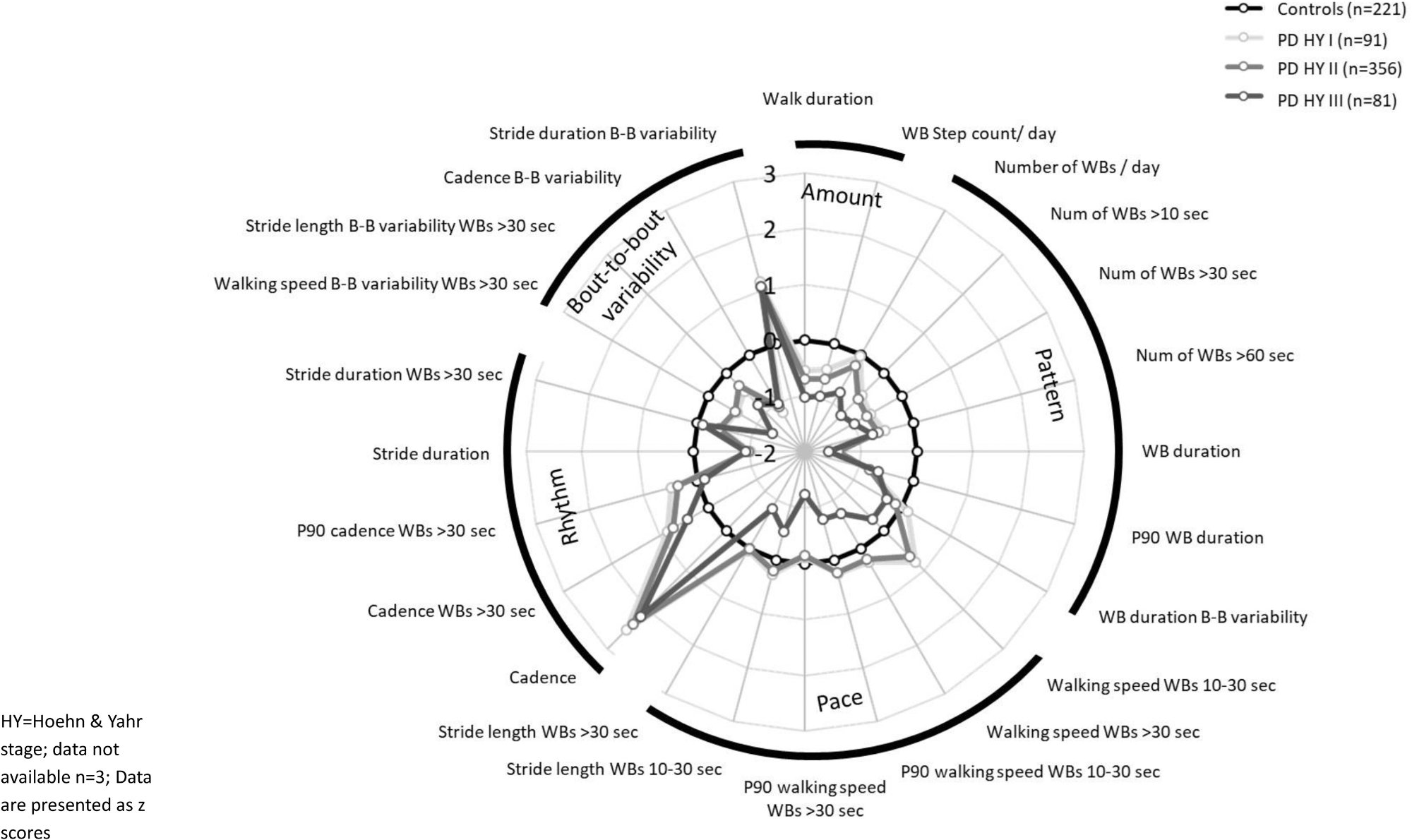
Figure 3 from Yarnall et al. Radar plot of digital mobility outcomes stratified by Hoehn and Yahr stage: controls (n=221), PD H&Y I (n=91), PD H&Y II (n=356), PD H&Y III (n=81). Graded separation is visible across walking activity, rhythm, and variability domains. Reproduced under CC BY-NC 4.0.
Stratifying by Hoehn and Yahr stage, the authors found significant differences in walking activity, rhythm, and bout-to-bout variability between healthy controls and mild disease (H&Y stage I–II), and between mild and moderate disease (H&Y stage III). Within the pace domain specifically, most differences emerged at the stage comparisons rather than in the overall PD-versus-control analysis. Patients at H&Y stage III walked less, at slower maximum speeds, with shorter stride lengths and lower bout-to-bout variability than those at H&Y stage I–II.
This graded separation across disease stages is precisely what a clinical trial endpoint needs to demonstrate. A disease-modifying therapy slows progression along that gradient. An outcome measure that tracks that gradient sensitively, capturing data continuously at home without requiring a clinic visit, offers something the MDS-UPDRS alone cannot.
The authors are explicit about what this study does and does not establish. It is a cross-sectional baseline characterization. It demonstrates that digital mobility outcomes discriminate between groups and across disease stages at a single point in time. Demonstrating that these outcomes are sensitive to treatment-related change over time requires longitudinal data from intervention trials, which the Mobilise-D consortium is positioned to generate as the cohort is followed.
Several technical caveats also merit attention. Walking speed and step counts in this study were lower than in some earlier PD cohorts, which the authors attribute to differences in walking bout definitions, algorithm validation approaches, and how weekly aggregates were calculated. These methodological details matter for comparing across studies and for pre-specifying endpoints in future trials. The authors have made their pipeline openly available, which is the right approach for a field that needs consistent measurement standards.
Site differences were observed, particularly at Tel Aviv, where PD participants had fewer and shorter walking bouts with slower speeds and shorter stride lengths compared to other sites. The authors discuss demographic and cultural factors as partial explanations. This variability across sites is a realistic feature of international trials and something future DMO-anchored studies will need to account for.
The regulatory context adds practical weight to these findings. The FDA and EMA have both engaged with the Mobilise-D data as part of formal pathways toward qualification of digital mobility outcomes as clinical trial endpoints. This study is part of a deliberate, methodical effort to build the evidentiary base that agencies require before these measures can anchor pivotal trials.
Gait and mobility are among the symptoms people with Parkinson's disease identify as most troubling and most limiting. The Mobilise-D study captures those symptoms as they are lived, across a full week of daily life, in over 600 people. That foundation, rigorous, large-scale, multi-site, and device-agnostic, is what the field needs to move real-world mobility from a compelling concept to a validated clinical trial endpoint.
Yarnall AJ, Alcock L, Schlenstedt C et al. "The future of clinical trials? Monitoring mobility as an outcome measure in Parkinson's disease: The Mobilise-D study." Journal of Parkinson's Disease 16(1), 110–124 (2026). https://doi.org/10.1177/1877718X251407026